Wegener’s granulomatosis (GPA); a scientific perspective.
Wegener’s granulomatosis (GPA or Granulomatosis with polyangiitis) is a multi-organ disease. Symptoms are various and the disease can affect the lungs, nose or eyes in addition to blood vessels, nerves and kidneys. However, if we are to understand the disease and how GPA evolves it is important to consider aspects of the disease independently – individuals with GPA may not have all of the symptoms.
It is common for individuals with GPA, especially in the early stages of disease, to suffer from soreness of the airways, nose or eyes, often for many years. We don’t know why this happens. The soreness is a consequence of acute inflammation at these sites. This inflammation is also often described as granulomatous giving the disease its name.
The other main group of symptoms is referred to as vasculitis, which means inflammation of the blood vessels. Vasculitis is caused by autoantibodies to neutrophil proteins that damage the small blood vessels and kidneys. There are several theories to explain how the damage happens, but we are not absolutely certain that any are entirely true. In terms of disease development, it is almost certain that vasculitis evolves after the acute inflammation in the upper airways, eyes or nose.
The St Thomas’ Wegener’s Trust is supporting research into understanding the inflammation around the nose and airways. Scientists at King’s College London are currently asking, “How does the autoantibody response that causes the vasculitis develop as a consequence of inflammation in the upper airways?” Understanding these processes may lead to new treatments for GPA.
Acute inflammation
Under normal circumstances, acute inflammation is the immune response that the body makes very rapidy when, for example, bugs such as bacteria cross a cut in the skin resulting in an infected wound. An infected wound can acquire pus, which is an accumulation of acute inflammatory cells such as neutrophils and macrophages. In health an infected wound that accumulates pus would resolve. For some reason the acute inflammation in GPA persists. We don’t know why the acute inflammation occurs or why it doesn’t resolve in GPA. There are reports in the scientific literature that suggest that the acute inflammation is a consequence of a long-term infection. However, this is not known for sure.
Granulomatous
The adjective granulomatous refers to the presence of granulomas in the affected tissues. Granulomas are clusters of macrophages associated together tightly to make a ball of cells. Granulomas are known to form in diseases caused by infection such as TB, but also in some other diseases that are not caused by infection such as Crohn’s disease in the gut. Therefore the presence of granulomas is difficult to interpret in terms of causes of disease.
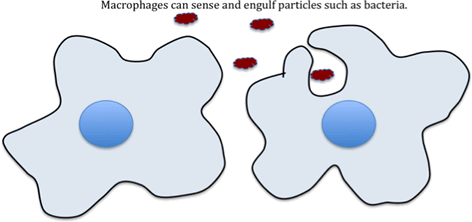
Macrophages
Macrophages are cells of the immune system that are present in most tissues of the body. They have a variety of different receptors on their surface that allow them to detect bugs such as bacteria. When they detect bugs they can surround them and engulf them. Once the macrophage has taken the bugs, it can kill them. The process of surrounding and engulfing is called ‘phagocytosis’.
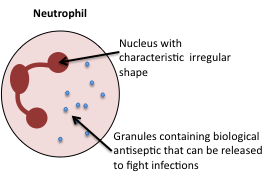
Neutrophils
Like macrophages, neutrophils can also detect bugs such as bacteria, using receptors on their surface. Like macrophages, they are also capable of phagocytosis. Unlike macrophages however, neutrophils have granules that they can release into tissues. The granules contain chemicals and molecules that function together as a very potent antiseptic.
Molecules on the surface of and inside neutrophils are common targets for autoantibodies in GPA. The autoantibodies to neutrophils are called termed ‘anti-neutrophil cytoplasmic antibody’ or ‘cANCA’ and the most common molecular target in GPA is called PR3 or proteinase 3.
Autoantibodies
Autoantibodies are antibodies that bind to components of human cells. In Wegener’s granulomatosis the autoantibodies bind to neutrophils, which in turn can bind to the lining of blood vessels and damage them; so called ‘vasculitis’.
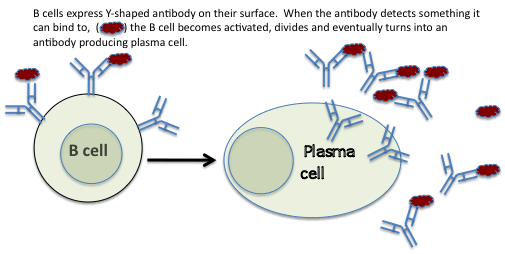
Antibodies and B cells
Antibodies are Y shaped molecules on the surface of the cells of the immune system called B cells. B cells are called B cells because they are made in the bone marrow. Antibodies on the surface of B cells bind to anything that enters the body using the shapes at the tips of the Y like a lock and key.
Virtually every B cell made in the bone marrow has a different shape at the tip of the Y so that no matter what enters the body, there will be a Y shape that fits it. If an antibody on the surface of a B cell binds something (could be an infection or a vaccine injected into the body) the B cell becomes activated and divides to increase the number of cells displaying antibody of the right shape. Eventually the B cell becomes a plasma cell that releases a lot of the antibody of the relevant shape to bind to and eliminate its target.
In health there are mechanisms that act as checkpoints to prevent B cells from making autoantibodies that bind to the body’s own cells. Exactly why or how B cells can start to make autoantibodies isn’t known.
Research funded by the St Thomas’ Wegener’s Trust.
Prof Jo Spencer and Prof David D’Cruz.
Our work involves the direct study of human cells and tissues with the approval of the local research ethics committee.
The Research Team, past and present.
We are focusing our research on two questions
1. New biological therapies are now available for the treatment of autoimmune conditions. Of particular interest to us, B cell depletion therapy (Rituximab) is effective for the treatment of Wegener’s granulomatosis (GPA). Our studies suggest that defects in T cell regulators of immune activity that are associated with disease can be resolved following rituximab therapy (see what we’ve found so far) and we are therefore focusing attention on this.
PhD student SangMi Kim is funded by the Wegener’s Trust to investigate functional T cell subsets in Wegener’s Granulomatosis under the supervision of Dr Susan John and Professor David D’Cruz. Their attention is focused on the T follicular helper cells; in particular how these cells respond differently in patients with Wegener’s granulomatosis. Dr Susan John is a T-cell expert who understands the chains of signals inside the cell and aims to unravel how and why the intracellular wiring is not correct in T cells of patients with Wegener’s granulomatosis.
2. Could a cheap, well-tolerated and readily available therapeutic used to treat other autoimmune diseases benefit patients with Wegener’s granulomatosis?
The Medical Research Council is funding a three year trial of hydrochloroquine in GPA (and other ANCA vasculitis). The Wegener’s Trust successfully laid the groundwork for the full clinical trial by funding Clinical Research Fellow Dr Alina Casian to develop the trial and undertake a smaller pilot study of 12 ANCA vasculitis patients which showed the benefits of hydroxychloroquine.
“If our study is positive, we hope that hydroxychloroquine, an old fashioned and safe drug widely used in other rheumatic conditions, will make a real difference to patients with this complex and challenging disease.” explains Professor David D’Cruz.
Hydroxychloroquine could be a safe and inexpensive game changer in controlling non-severe ANCA vasculitis, reducing the reliance on steroids and the need for toxic steroid sparing agents like cyclophosphamide or rituximab, as well as reducing associated complications such as infections, blood clots and stiffness of blood vessels. This HAVEN trial is to address the surprising lack of studies of hydroxychloroquine in ANCA vasculitis given its long history of successful use in other auto-immune conditions. Quinine was first used for patients with Lupus by Reverend Payne at St Thomas’ in 1894, and hydroxychloroquine has also been a safe, successful treatment for rheumatoid arthritis for over 50 years.